OUR RESEARCH
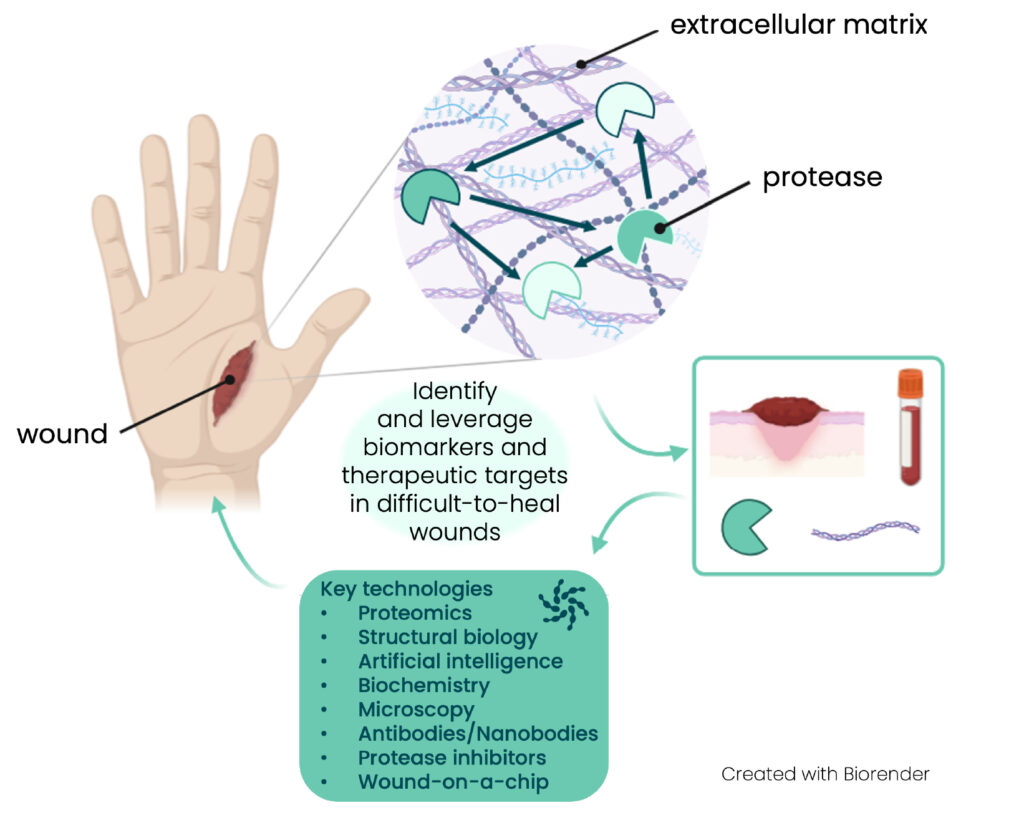
Understanding and targeting extracellular matrix remodelling to improve wound healing.
| WORK PACKAGE 1
Proteolytic networks in normal and pathological wounds
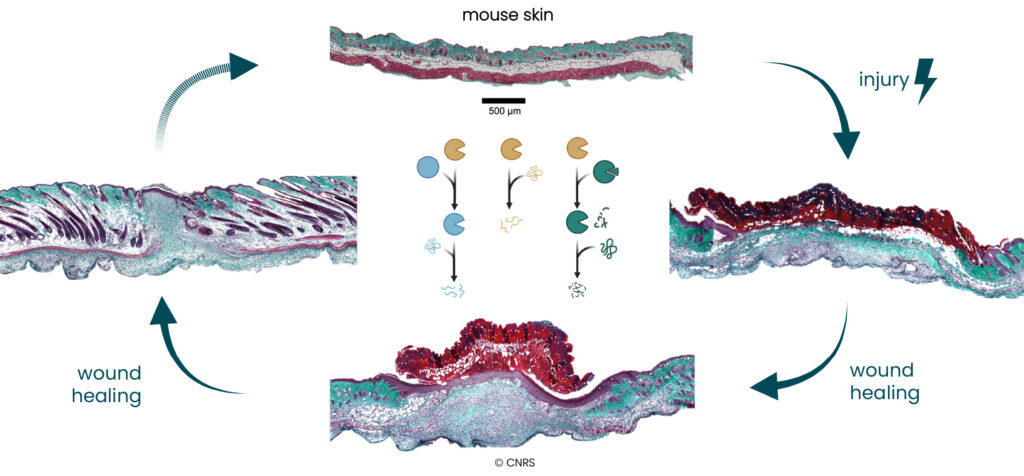
Doctoral candidates (DCs) 2, 3a, 3b and 4 will study different types of clinical samples and relevant mouse models, previously developed by consortium partners. They will focus on a selection of representative sample types corresponding to acute wounds (snake bites, surgery wounds), chronic wounds (venous leg ulcers, epidermolysis bullosa (EB) wounds), dermonecrotic wounds (snake bites) and fibrotic wounds (advanced stages of EB and dermonecrotic wounds). Besides tissue histology and targeted immuno-assays, proteolytic networks will be revealed through high-end proteomic approaches, including extensive analysis of protein neo-N-termini and computational approaches to connect proteases with their extracellular matrix substrates (DC-3b, DC-4). Mouse models and 3D organotypic skin cultures will also be used to analyse already-identified relevant protease targets and validate the new targets that will emerge from the proteomic data (DC-2, DC-3a).
| WORK PACKAGE 2
Biochemical and structural properties of remodelling proteases, their extracellular matrix substrates and their endogenous inhibitors
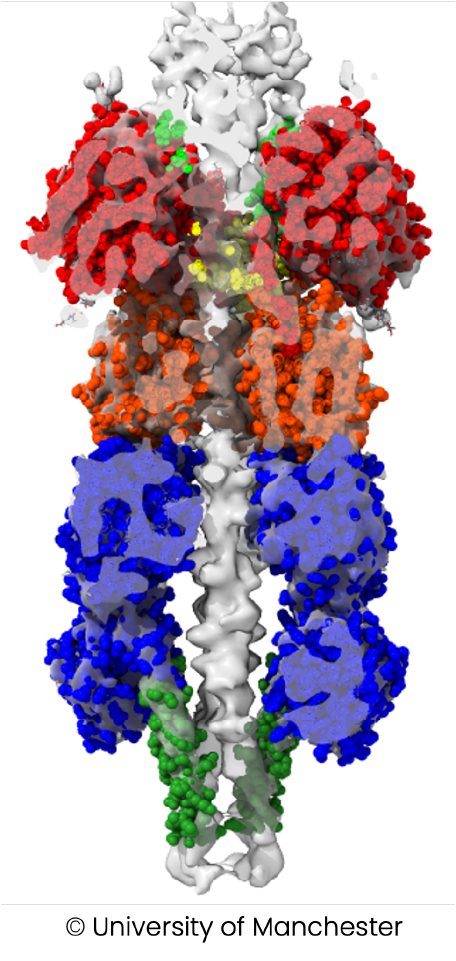
Promising protease targets identified from previous work of consortium partners or supported by literature will be studied at the molecular level, along with their extracellular matrix substrates and their endogenous inhibitors, to get a better understanding of their structure-function relationships. Mechanisms of action and structures of protease-substrate or protease-inhibitor complexes will be analysed by DC-1, DC-5 and DC-6 using advanced biochemical and biophysical methods, X-ray crystallography or cryo-electron microscopy. These results will also serve as a basis for the development of therapeutic molecules. DC-7 will focus on collagen as a substrate and exploit the fact that collagenase activity and excessive collagen degradation are excellent biomarkers of tissue damage to develop a novel collagen-based model substrate and fluorescent probe.
| WORK PACKAGE 3
Therapeutic impact: detecting and normalizing protease activities in wounds
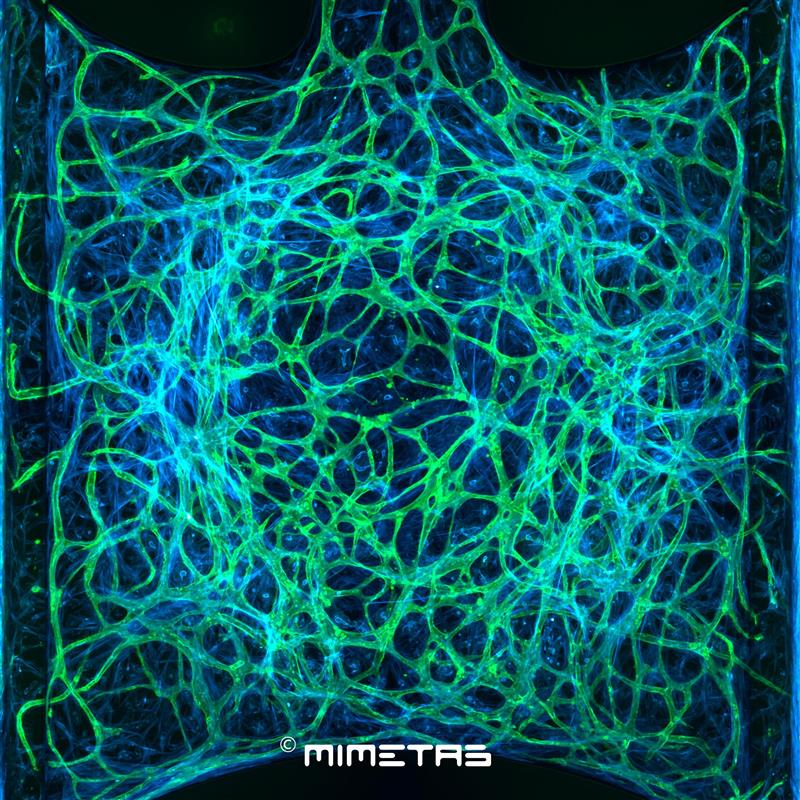
This WP deals with the most direct translational aspects of our work. DC-8 will develop single-domain antibodies (Nanobodies®) to inhibit or detect, after labelling, selected metalloprotease targets characterized in Workpackages 1-2. The substrate specificity and preference for unnatural amino-acids of serine or cysteine protease targets will also be studied by DC-9 in order to develop a completely original series of macrocyclic inhibitory peptides. DC-10 will develop an in vitro 3D skin model and validate its relevance to study wounding, especially the recruitment of immune cells which is difficult to model in vitro, and to test anti-protease drugs. Finally, DC-11 will build on the ECM fragments identified in Workpackages 1-2 to select relevant biomarker assays in the vast biomarker portfolio that is already available in the consortium, develop new biomarker assays according to the needs and validate them using human and mouse samples.